A monumental pre-print research paper published on Monday by the Cleveland Clinic, MedRxiv uncovered that MRNA vaccines can enhance your likelihood of getting COVID-19, and any extra booster shots will only further this risk. Astonishingly, those who remain unvaccinated have the most minimal probability of contracting coronavirus compared to any other groups. Despite the slight risk involved, it is nevertheless counter to how government authorities have advertised and legislated for mass vaccinations. Individuals who received more than three vaccines had the greatest risk.
Cleveland Clinic researchers Nabin K. Shrestha, Patrick C. Burke, Amy S. Nowacki, James F. Simon and Amanda Hagen as well as Steven M Gordon reported on the study’s results.
“The association of increased risk of COVID-19 with higher numbers of prior vaccine doses in our study, was unexpected.”
Quote from the study\
In the autumn, 51,011 Cleveland Clinic employees participated in a study to analyze how successful the new bivalent MRNA vaccine would be at shielding them from infections. Disconcertingly, a mere 21 percent of employees received the bivalent booster which was only proven to possess 30 percent efficacy. All told, 5% of employees in the study tested positive for COVID-19 during their 13 week period.
Vaccines have produced varied outcomes. Those in the elderly, immuno-compromised and comorbid groups have observed lowered hospitalizations and fatalities due to vaccinations. On the other hand, those who are younger and healthier may experience side effects such as myocarditis, clotting issues or menstrual irregularities. This study adds to the information about the vaccines.
Excerpts
This article is a preprint and has not been certified by peer review [what does this mean?]. It reports new medical research that has yet to be evaluated and so should not be used to guide clinical practice.
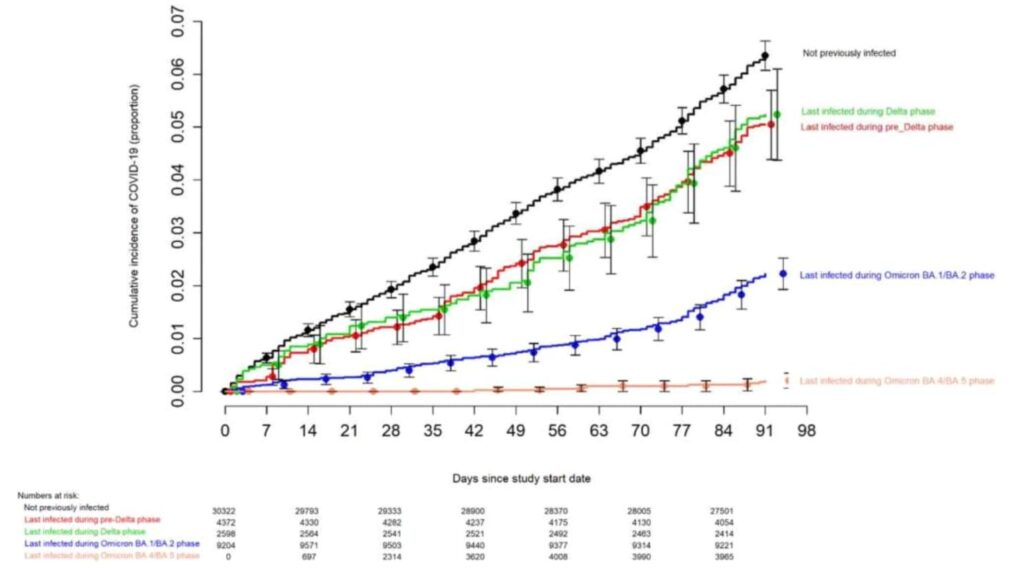
ABSTRACT Background The purpose of this study was to evaluate whether a bivalent COVID-19 vaccine protects against COVID-19. Methods Employees of Cleveland Clinic in employment on the day the bivalent COVID-19 vaccine first became available to employees, were included. The cumulative incidence of COVID-19 was examined over the following weeks. Protection provided by vaccination (analyzed as a time-dependent covariate) was evaluated using Cox proportional hazards regression. The analysis was adjusted for the pandemic phase when the last prior COVID-19 episode occurred, and the number of prior vaccine doses received. Results Among 51011 employees, 20689 (41%) had had a previous documented episode of COVID-19, and 42064 (83%) had received at least two doses of a COVID-19 vaccine. COVID-19 occurred in 2452 (5%) during the study. Risk of COVID-19 increased with time since the most recent prior COVID-19 episode and with the number of vaccine doses previously received. In multivariable analysis, the bivalent vaccinated state was independently associated with lower risk of COVID-19 (HR, .70; 95% C.I., .61-.80), leading to an estimated vaccine effectiveness (VE) of 30% (95% CI, 20-39%). Compared to last exposure to SARS-CoV-2 within 90 days, last exposure 6-9 months previously was associated with twice the risk of COVID-19, and last exposure 9-12 months previously with 3.5 times the risk. Conclusions The bivalent COVID-19 vaccine given to working-aged adults afforded modest protection overall against COVID-19, while the virus strains dominant in the community were those represented in the vaccine. Summary Among 51011 working-aged Cleveland Clinic employees, the bivalent COVID-19 vaccine booster was 30% effective in preventing infection, during the time when the virus strains dominant in the community were represented in the vaccine. INTRODUCTION When the original Coronavirus Disease 2019 (COVID-19) vaccines first became available in 2020, there was ample evidence of efficacy from randomized clinical trials [1,2].Vaccine effectiveness was subsequently confirmed by clinical effectiveness data in the real world outside of clinical trials [3,4], including an effectiveness estimate of 97% among employees within our own healthcare system [5]. This was when the human population had just encountered the novel Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus, and the pathogen had exacted a high burden of morbidity and mortality across the world. The vaccines were amazingly effective in preventing COVID-19, saved a large number of lives, and changed the impact of the pandemic. Recognition that the original COVID-19 vaccines provided much less protection after the emergence of the Omicron variant, spurred efforts to produce newer vaccines that were more effective. These efforts culminated in the approval by the US Food and Drug Administration, on 31 August 2022, of bivalent COVID-19 mRNA vaccines, which contained antigens represented in the original vaccine as well as antigens representing the BA.4/BA.5 lineages of the Omicron variant. Given the demonstrated safety of the earlier mRNA vaccines and the perceived urgency of need of a more effective preventive tool, these vaccines were approved without demonstration of effectiveness in clinical studies. The purpose of this study was to evaluate whether the bivalent COVID-19 vaccine protects against COVID-19. Since the arrival of the COVID-19 pandemic at Cleveland Clinic in March 2020, employee access to testing has been a priority. Systems were designed to enable Occupational Health to interview and remotely monitor symptoms for all employees while the latter were isolated at home. Voluntary vaccination for COVID-19 began on 16 December 2020, and the monovalent vaccine as a booster became available to employees on 5 October 2021. The bivalent COVID-19 vaccine began to be offered to employees on 12 September 2022. This date was considered the study start date. OUTCOME The study outcome was time to COVID-19, the latter defined as a positive NAAT for SARS-CoV-2 any time after the study start date. Outcomes were followed until December 12, 2022. RESULTS Of 51977 eligible subjects, 966 (1.9%) were excluded because of missing age or gender. Of the remaining 51011 employees included in the study, 34507 (68%) had been in employment since before the onset of the COVID-19 pandemic (pre-pandemic hires). 1794 subjects (3.5%) were censored during the study period because of termination of employment before the end of the study. By the end of the study, 10804 (21%) were bivalent vaccine boosted. The bivalent vaccine was the Pfizer vaccine in 9595 (89%) and the Moderna vaccine in the remaining 1178. Altogether, 2452 employees (5%) acquired COVID-19 during the 13 weeks of the study. RISK OF COVID-19 BASED ON PRIOR INFECTION AND VACCINATION HISTORY The risk of COVID-19 varied by the phase of the epidemic in which the subject’s last prior COVID-19 episode occurred. In decreasing order of risk of COVID-19 were those never previously infected, those last infected during the pre-Delta or Delta phase, those last infected during the Omicron BA.1/BA.2 phase, and those last infected during the Omicron BA.4/BA.5 phase (Figure 1).
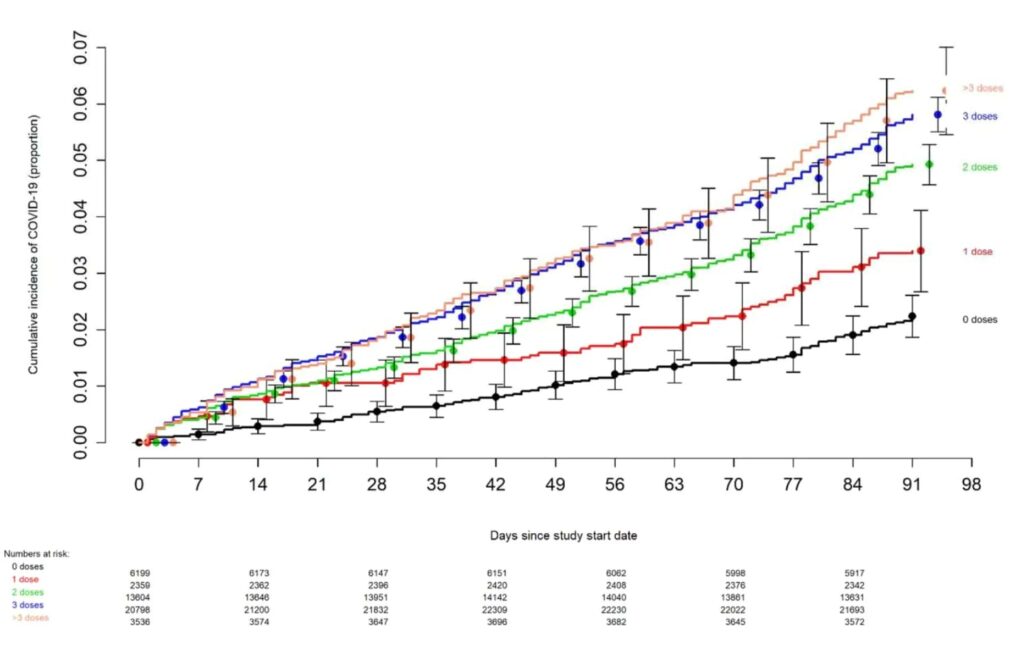
BRAKE FOR IT The risk of COVID-19 also varied by the number of COVID-19 vaccine doses previously received. The higher the number of vaccines previously received, the higher the risk of contracting COVID-19 (Figure 2).
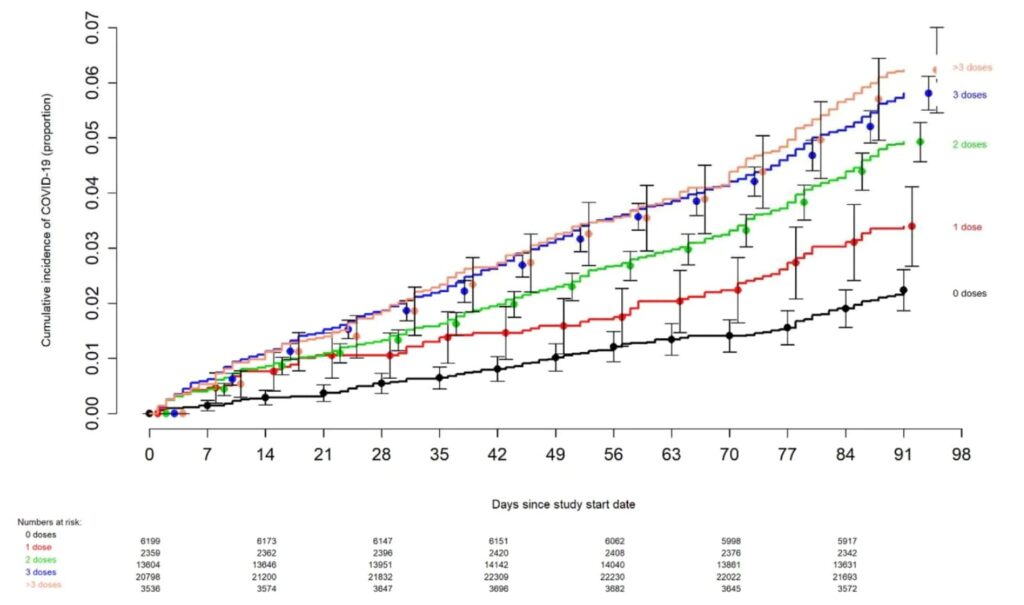
…The multivariable analyses also found that, the more recent the last prior COVID-19 episode was the lower the risk of COVID-19, and that the greater the number of vaccine doses previously received the higher the risk of COVID-19. …DISCUSSION This study found that the current bivalent vaccines were about 30% effective overall in protecting against infection with SARS-CoV-2, when the Omicron BA.4/BA.5 lineages were the predominant circulating strains. The magnitude of protection afforded by bivalent vaccination was similar to that estimated in a recent study using data from the Increasing Community Access to Testing (ICATT) national SARS-CoV-2 testing program [16]. The evolution of the SARS-CoV-2 virus necessitates a more nuanced approach to assessing the potential impact of vaccination than when the original vaccines were developed. Additional factors beyond vaccine effectiveness need to be considered. The association of increased risk of COVID-19 with higher numbers of prior vaccine doses in our study, was unexpected. A simplistic explanation might be that those who received more doses were more likely to be individuals at higher risk of COVID-19. A small proportion of individuals may have fit this description. However, the majority of subjects in this study were generally young individuals and all were eligible to have received at least 3 doses of vaccine by the study start date, and which they had every opportunity to do. Therefore, those who received fewer than 3 doses (>45% of individuals in the study) were not those ineligible to receive the vaccine, but those who chose not to follow the CDC’s recommendations on remaining updated with COVID-19 vaccination, and one could reasonably expect these individuals to have been more likely to have exhibited higher risk-taking behavior. Despite this, their risk of acquiring COVID-19 was lower than those who received a larger number of prior vaccine doses. This is not the only study to find a possible association with more prior vaccine doses and higher risk of COVID-19. A large study found that those who had an Omicron variant infection after previously receiving three doses of vaccine had a higher risk of reinfection than those who had an Omicron variant infection after previously receiving two doses of vaccine [21]. Another study found that receipt of two or three doses of a mRNA vaccine following prior COVID-19 was associated with a higher risk of reinfection than receipt of a single dose [7]. We still have a lot to learn about protection from COVID-19 vaccination, and in addition to a vaccine’s effectiveness it is important to examine whether multiple vaccine doses given over time may not be having the beneficial effect that is generally assumed. In conclusion, this study found an overall modest protective effect of the bivalent vaccine booster against COVID-19, among working-aged adults. The effect of multiple COVID-19 vaccine doses on future risk of COVID-19 needs further study.
(END OF REPORT)
The current FDA recommendations updated as of December 9, 2022:
On December 9th, 2022, the CDC made an important move to expand access for updated (bivalent) COVID-19 vaccines in children aged 6 months through 5 years. For more information on this exciting development and its implications, please consult both the CDC and FDA media statements.

