On a misty Seattle morning, Carolina Reid was one of ten volunteers sitting in a room, half-asleep as they waited to take part in a clinical trial for an unproven malaria vaccine.
When it was Reid’s turn, she put her arm over a cardboard box filled with 200 mosquitoes. The box had mesh that kept the mosquitoes in but still let them bite. A scientist then covered her arm with a black cloth because mosquitoes like to bite at night.
Hundreds of mosquitoes descended upon her arm & the feeding frenzy began.
“My whole forearm swelled and blistered,” says Reid. “My family was laughing, asking like, ‘why are you subjecting yourself to this?'” And she didn’t just do it once. She did it five times.
You may be thinking this is a joke?
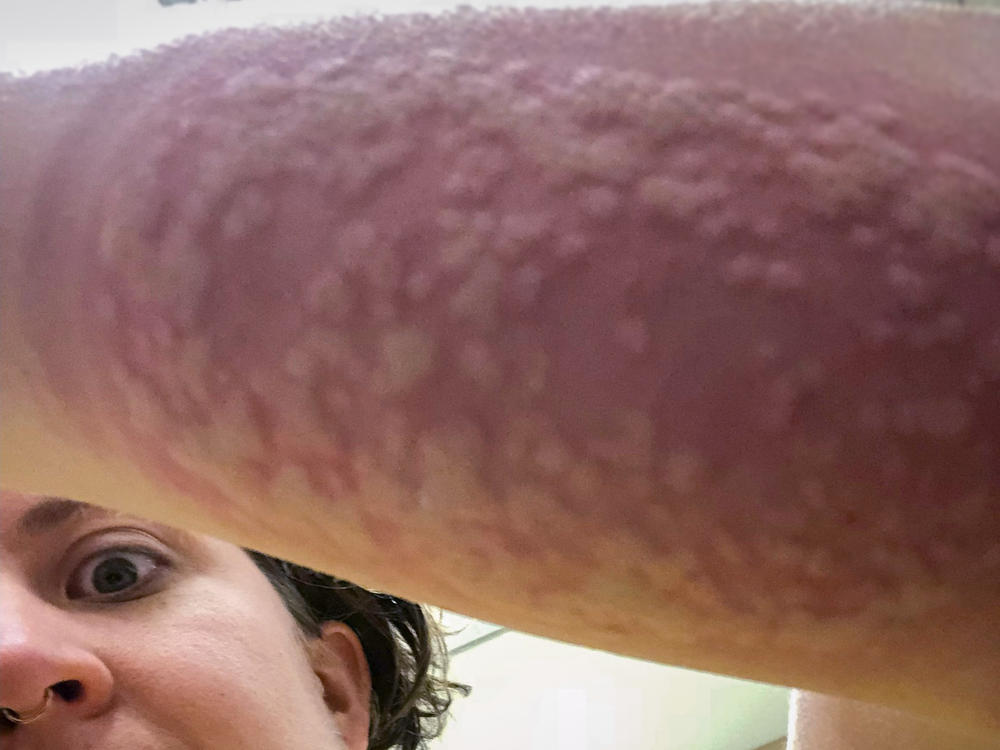
But it’s not.
“We use the mosquitoes like they’re 1,000 small flying syringes,” explains Dr. Sean Murphy, a University of Washington, Seattle physician and scientist who was the lead author on a paper in Science Translational Medicine released on August 24, details the vaccine trials.
The insects transmit live malaria-causing Plasmodium parasites that have been genetically modified not to make people sick. The body still produces antibodies against the weakened parasite so it is prepared to fight the real thing.
To be clear, Murphy isn’t proposing to immunize millions of people with mosquitoes (supposdily). Mosquitoes have previously been used in clinical trials to deliver malaria vaccines, but this isn’t often done.

He and his coworkers took this path since it’s more expensive and time-consuming to develop a parasite formulation that may be injected. The parasites mature within mosquitoes, so at this proof of concept stage – as early-phase trials are known – it makes sense to employ them for delivery.
“They went old school with this one,” says Dr. Kirsten Lyke, a physician and vaccine researcher at the University of Maryland School of Medicine who was not involved in the study. “All things old become new again.”
She refers to the use of a genetically modified live parasite as “a total game changer” for vaccine development.
Although this kind of vaccination is still in its infancy, and this vaccine is not yet perfect, a trial of 26 people showed that the modified parasites were able to protect some participants from malaria for a few months.
According to Murphy, this technique might one day lead to a more effective vaccine than the worlds first malaria vaccine, RTS,S developed by GlaxoSmithKline. It was given the green light by the World Health Organization last year, but its effectiveness rate is only 30-40 percent.
Mosquitoes and malaria – a toxic relationship
In 2018, Reid was unemployed and looking for work when she saw an ad seeking participants for a clinical trial that would pay $4,100. Though the money was enticing, what ultimately motivated her to join the trial was speaking with friends who had contracted malaria. They helped her see that participating in important research could be much more rewarding than simply earning a paycheck.
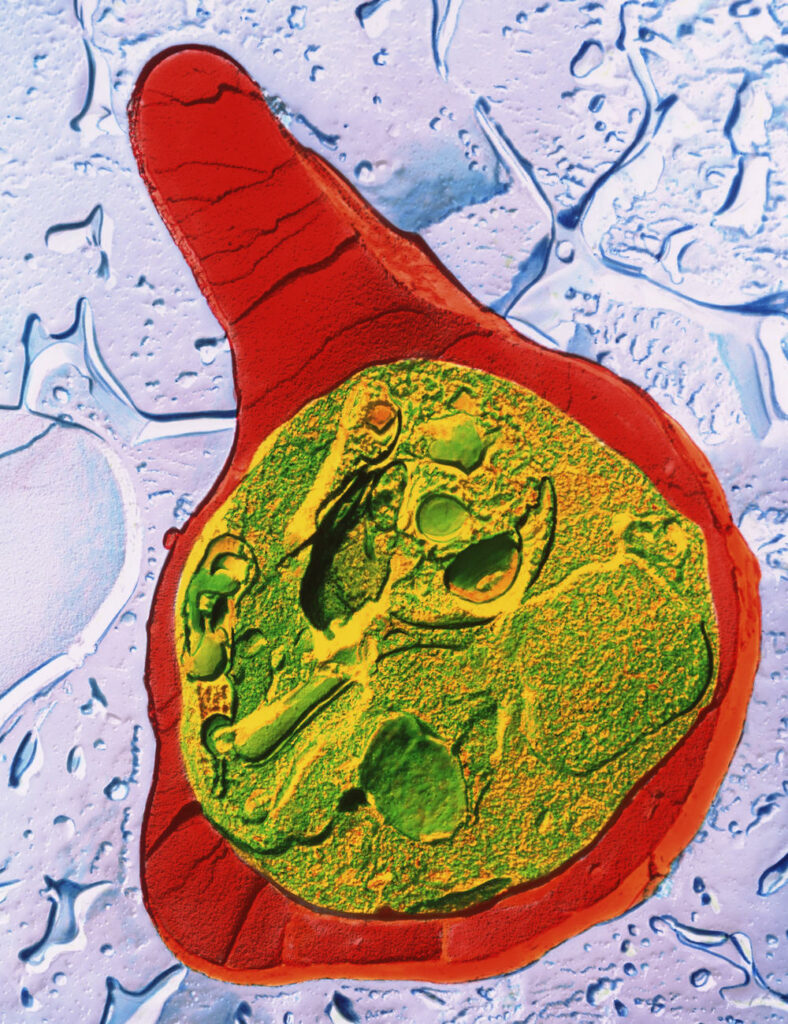
Malaria parasites call the salivary glands of Anopheles mosquitoes their home. The warm climate in Africa provides optimal growing conditions for the parasite, making it most prevalent there. To contract malaria, people must be bitten by an infected mosquito – someone who is already carrying the disease-ridden parasite. If a person has contracted malaria, they can infect future bites from other mosquitoes and continue spreading the disease this way.
Mosquito netting, insecticidal sprays, anti-malarial medicines, and even genetically modified mosquitoes that can’t bite or lay eggs have all been used to combat malaria.
Even with those measures, scientists estimate there are over 240 million cases of malaria a year and over 600,000 deaths – which is why vaccines are needed, they say.
A promising start – but there’s room for improvement
Murphy believes that this experimental vaccine will create a stronger response from the immune system than the currently approved RTS,S vaccine. The current WHO-approved version only targets “one out of more than 5,000 proteins” whereas Murphy’s proposed method uses an entire weakened parasite.
This team is not the first to develop a malaria vaccine from disarmed parasites, but they are the first to use CRISPR technology.
To see if the method was successful, Reid and the other participants had to get bitten by mosquitoes carrying the malaria parasite.
Out of 14 persons who were given a vaccination against malaria, seven became sick, indicating that the vaccine was only 50% effective. The other seven had no more than a few months’ protection.
“I actually cried when they told me I had malaria because I developed such a close relationship with the nurses,” Reid says. Even though she was determined to see the trials through, her malaria made her ineligible. After being given a medication to clear the infection, she was sent home.
“We think we can obviously do better,” says Stefan Kappe, an author of the study and parasitologist at University of Washington Seattle and Seattle Children’s Research Institute. He and Murphy are currently working on a project to improve the vaccine’s effectiveness by encapsulating it in syringes rather than using mosquitoes, allowing them to deliver the dosage precisely. A higher initial dose might result in greater protection for a longer duration of time.
According to Lyke, some scientists believe that using a slightly more developed variety of the parasite than the one in this vaccine may allow the body additional time to build an immune response. The team is already working on it, according to Kappe.
There are a lot of questions to consider if future trials are successful. To begin with, how much would this sort of vaccine cost? The researchers are working with a boutique firm called Sanaria to create the modified parasites. Increasing manufacturing capability in order to scale up production will need investment, according to Kappe.
As for Reid, her experience was so positive that she went on to participate in clinical trials for a bird flu vaccination and the Moderna COVID-19 vaccine. She says she will continue to enroll in vaccine clinical trials “for the rest of my life.”

An impressive share, I just given this onto a colleague who was doing a little analysis on this. And he in fact bought me breakfast because I found it for him.. smile. So let me reword that: Thnx for the treat! But yeah Thnkx for spending the time to discuss this, I feel strongly about it and love reading more on this topic. If possible, as you become expertise, would you mind updating your blog with more details? It is highly helpful for me. Big thumb up for this blog post!